What's New Archive
It is not unusual to experience mild to moderate pain after dental procedures. Our recommendations for pain control after treatment are as follows:
Our Philosophy on
Dental Implants
We have been restoring dental implants in our office for over twenty years and have completed over one thousand restorations with very good success. We collaborate with the best implant surgeons in our area, the best laboratories, and the best implant companies. Our cases are planned very carefully and take time to execute. With this meticulous process, we rarely see complications. While some dentists advertise low-cost and same-day implants we feel there are increased risks and complications in using off-brand implants and proceeding too quickly. In order to be highly predictable, dental implants need to be well integrated into the bone, with stable tissue around them, prior to placing the final restorations. Dental implants can be used in many situations: (1) to replace a single missing tooth, (2) to replace multiple missing teeth, (3) to support a removable denture that would be unstable without this additional support. The advantage to using a dental implant to replace a missing single tooth is highest when the adjacent teeth do not need any restorations or the adjacent teeth are not strong enough to support a traditional fixed bridge. Patients that have removable dentures that are not stable often can be helped with implants to support these appliances or eliminate them completely.
Tooth Wear
The following information is from a review by Robyn R. Loewen, D.D.S., Robert J. Marolt, D.D.S., John D. Ruby, D.M.D., Ph.D.
Tooth wear can be caused by erosion, attrition, and abrasion.
Abrasion loss of tooth structure by contact between tooth and another source, usually without any evidence of decay or decalcification. (i.e. toothbrush abrasion).
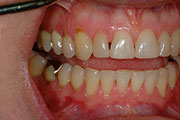
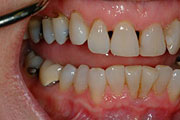
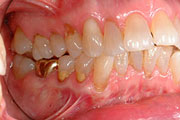
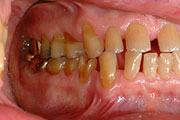
Attrition loss of tooth structure from tooth-to-tooth contact (i.e. tooth grinding).
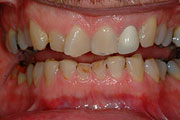
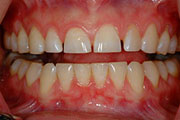
Erosion loss of tooth structure by a chemical substance (i.e. acid).
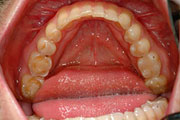
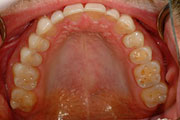
Gastroesophageal reflux disease (GERD) and frequent vomiting cause the loss of enamel on teeth. The causative agents of erosion are usually acidic substances, whether intrinsic (gastric acid from reflux or repeated vomiting) or extrinsic (consumption of acidic foods or beverages, repeated exposure to chlorinated water, and industrial chemical exposure.
The Truth Behind Sour Candies
- Sour candies are very acidic, with a low pH.
- Acid weakens and wears away tooth enamel.
The Signs of Tooth Erosion
- Sensitivity may occur when tooth enamel wears away.
- The discoloration is visible as a slightly yellow appearance on the tooth crown or root surface.
- The translucency of the front teeth may appear along the biting edges.
- Rounding of teeth occurs along the surfaces and edges of the teeth.
- Cracks and roughness appear along the edges of the teeth.
- Cupping (which appears like potholes in the tooth) develops on the chewing surfaces of the teeth. At this severe stage, fillings may actually appear to stand proud.
How to Protect Your Teeth
- Reduce or eliminate consumption of sour candies.
- If you do eat sour candy, swish your mouth with water, drink milk, or eat cheese afterward to neutralize the acids.
- Chew sugar-free gum to produce saliva which protects tooth enamel. We recommend Sugar-free gum with Xylitol (I.e. Hershey’s Ice Breakers).
- After eating sour candy or other acidic foods or drinks, wait one hour before brushing your teeth. Brushing right away increases the harmful effects of acid on teeth.
- Ask us about ways to reduce sensitivity or minimize enamel loss if erosion has begun.
Diet and Hygiene Modifications
Every attempt should be made to reduce the frequency of consumption of acidic candies, and such foods should be restricted to main meals. For example, a change in the timing of eating sour candy between meals or at bedtime to mealtime can reduce the severity of the acid attack due to more efficient clearance time and the buffering benefits of saliva. Patients are encouraged to finish a meal with something neutral or alkaline — cheese, milk, or sugar-free chewable antacid tablets. Chewing sugar-free gum will stimulate saliva flow, providing a natural buffering action. Rubbing bicarbonate containing toothpaste on the teeth with a fingertip will reduce the acid challenge on the tooth surface. Also, rinsing with water rather than brushing teeth immediately following an acid challenge can reduce demineralization by clearing the acid from the oral cavity, and will prevent inordinate damage by toothbrush abrasion of the fragile enamel surface. Finally, patients should use less abrasive “sensitive” toothpaste or those with bicarbonate as an active ingredient, rather than highly abrasive whitening toothpastes.
Restoration
The goal of restorative treatment should be to maintain adequate function and esthetics of primary and permanent teeth. Erosion from the chewing or sucking of acidic candy primarily affects the top surfaces of the posterior teeth. Eroded primary teeth should be restored to maintain bite opening and tooth size, reduce symptomatic sensitivity, and provide nerve protection for the maintenance of vitality until the tooth is lost. Frequently, full-coverage stainless steel crowns are the most appropriate restorative choice for severely eroded posterior primary teeth.
The restoration of eroded permanent teeth requires choosing the least invasive procedure while providing maximum protection from the further loss of tooth structure. Sealants and resin-bonded restorations in “cupped” cusp tip lesions will provide mechanical protection for the affected surfaces, reduce dentin hypersensitivity, and improve the appearance of severely eroded posterior teeth. Many of these teeth will eventually require crowns to preserve the remaining tooth structure and provide adequate esthetics and function. Since full-coverage restorations are costly and involve significant additional loss of tooth structure, it is essential that appropriate early counseling and behavior modification should occur to prevent the need for such invasive treatment.
Summary
- The acid in soft drinks, whether they contain sugar or not, is the primary cause of weakening tooth enamel.
- The acid attacks your teeth. Each acid attack lasts about 20 minutes.
- The acid attack starts over again with every sip.
- Ongoing acid attacks weaken your tooth enamel.
- Bacteria in your mouth cause cavities when tooth enamel is damaged.
- If you have a receding gum line, acid does more damage below the gum line than above it. This is particularly a concern for adults.
- Drink soft drinks in moderation.
- Use a straw to keep the sugar away from your teeth.
- After drinking, swish your mouth out with water to dilute the sugar.
- Never drink soft drinks or juice before bedtime because the liquid pools in your mouth and coats your tongue and teeth with sugar and acid.
- Drink water instead of soft drinks. It has no sugar, no acid, and no calories.
- Get regular checkups and cleanings to remove bacteria buildup (plaque). Floss, too.
- Use fluoride toothpaste to protect your teeth.
Data courtesy of Dr. John Ruby, University of Alabama Birmingham School of Dentistry, 2007. Also, most of this data can be found on the Minnesota dental association’s website.
Why is fluoride NOW in Newport Beach water?
About Fluoride
Fluoride helps teeth resist decay by strengthening the protective layer of tooth enamel, and can reverse newly formed cavities. Fluoride has been added to U.S. drinking water supplies since 1945. More than two-thirds of the population in the U.S. are served by public water systems that are optimally fluoridated.
Fluoridation
Starting November 19, 2007, Metropolitan Water District (MWD) began adding fluoride to the water that it serves to Newport Beach. In line with recommendations from the California Department of Public Health, as well as the U.S. Centers for Disease Control and Prevention, MWD will adjust the natural fluoride level in the water, which ranges from 0.1 to 0.4 parts per million, to 0.7 to 0.8 parts per million. Currently, the City of Newport Beach receives approximately 18% of its water from MWD and 82% from its groundwater well sources. The City of Newport Beach will not be adding fluoride to its well sources. The City of Newport Beach’s groundwater well sources contains a natural occurrence of fluoride Also beginning this November, the City of Newport Beach will begin a fluoride monitoring program to monitor the levels of fluoride throughout the distribution system.
Fluoridated Water and Supplements
Drinking optimally fluoridated water on a regular basis makes the use of fluoride tablets or drops unnecessary. However, the continued use of fluoride treatments by professional dental caregivers is recommended. Drinking fluoridated water should be part of a total treatment plan for healthy gums and teeth, combined with brushing and flossing your teeth regularly and using less sugar in your diet. People who do not wish to drink fluoridated water should know that most bottled waters do contain levels of fluoride below the optimum range.
Here are some answers to questions we often get asked
Q. How does fluoride protect teeth against tooth decay?
Tooth decay occurs when acids produced by bacteria in the mouth dissolve or demineralize the teeth. Fluoride protects against tooth decay by slowing down or stopping demineralization, promoting remineralization, and keeping the bacteria from producing too much acid. Technically fluoride reduces the solubility of teeth by changing out a hydroxyl group for a Fluoride ion.
Q. Is fluoride harmful to my health?
There have been thousands of studies that have looked at whether fluoride in drinking water is harmful to human health. These studies have looked at whether there is a link between fluoride and cancer, bone fractures as well as adverse effects on the immune system, kidneys, digestive system and reproductive system. The American Dental Association, in reviewing the body of scientific evidence, concluded that the overwhelming evidence indicates that fluoridation of community water supplies is both safe and effective.
Q. Will I miss the benefits from fluoridation if I drink bottled water, vended water or water from a store or a home filtration system?
If you mostly drink bottled water, water from vending machines, or water from water stores, you may miss the benefits of an optimally fluoridated water supply. If you use vended water or purchase water from a water store and the water is likely treated by reverse osmosis or distillation, the water is most likely low in fluoride.
Q. Should I give fluoridated water to my infant?
A National Research Council study has raised the possibility that infants could receive a greater than the optimal amount of fluoride. A liquid concentrate of powdered baby formula that has been mixed with water containing fluoride during a time that they are developing teeth may cause them to be more susceptible to enamel fluorosis. The American Dental Association has issued interim guidance on fluoride intake for infants and young children. Among the recommendations are that if liquid concentrate or powdered infant formula is the primary source of nutrition for infants, it can be mixed with water that is fluoride-free or contains low levels of fluoride to reduce the risk of fluorosis.
Q. Will fluoridated water harm my pets?
No evidence exists that indicates fluoridated water at the levels prescribed for human consumption is harmful to animals or pets.
Bisphosphonates
Bisphosphonates (i.e. Fosamax, Actonel, Boniva) and their role in bone death.
If you use a bisphosphonate medication for osteoporosis or as part of cancer treatment therapy you should advise your dentist. In fact, any time your health history or medications change, you should make sure the dental office has the most recent information in your patient file. The American Dental Association recommends that a patient be examined by a dentist prior to initiating bisphosphonate therapy to reduce further complications.
Here's why
Some bisphosphonate medications (such as Fosamax, Actonel, Boniva) are taken orally to help prevent or treat osteoporosis and Paget’s disease of the bone. Others, such as Aredia, Bonefos, Didronel, or Zometa, are administered intravenously as part of cancer therapy to reduce bone pain associated with metastatic breast cancer, prostate cancer, and multiple myeloma.
In rare instances, some individuals receiving intravenous bisphosphonates for cancer treatment have developed osteonecrosis of the jaw, a rare but serious condition that involves severe loss, or destruction, of the jawbone.
Symptoms of Osteonecrosis of the jaw include, but are not limited to:
Pain, swelling, or infection of the gums or jaw
Gums that are not healing
Loose teeth
Numbness or a feeling of heaviness in the jaw
Drainage
Exposed bone
The following information comes directly from the American Dental Association and can be found on their website at www.ada.org
Overview
Reports of bisphosphonate-associated osteonecrosis of the jaw (BON) associated with the use of Zometa (zoledronic acid) and Aredia (pamidronate) began to surface in 2003. The majority of reported cases have been associated with dental procedures such as tooth extraction; however, less commonly BON appears to occur spontaneously in patients taking these drugs. Zoledronic acid and pamidronate are intravenous (i.v.) bisphosphonates used to reduce bone pain, hypercalcemia, and skeletal complications in patients with multiple myeloma, breast, lung, and other cancers and Paget’s disease of bone.
Several cases of BON have also been associated with the use of the oral bisphosphonates, Fosamax (alendronate), Actonel (risedronate), and Boniva (ibandronate), for the treatment of osteoporosis; however, it is not clear if these patients had other conditions that would put them at risk for developing BON.
The table below lists all oral and i.v. bisphosphonates currently on the market in the U.S.
Orally Administered Bisphosphonates
Intravenously Administered Bisphosphonates
Clinical Presentation
The typical clinical presentation of BON includes pain, soft-tissue swelling, and infection, loosening of teeth, drainage, and exposed bone. These symptoms may occur spontaneously, or more commonly, at the site of previous tooth extraction. Patients may also present with feelings of numbness, heaviness, and dysesthesias of the jaw. However, BON may remain asymptomatic for weeks or months, and may only become evident after finding an exposed bone in the jaw.
Dental Management
It is important to understand that, based on the information currently available, the risk for developing BON is much higher for cancer patients on i.v. bisphosphonate therapy than the risk for patients on oral bisphosphonate therapy. Therefore, there are different recommendations for the dental management of these patients.
For patients on oral bisphosphonate therapy
The risk of developing BON in patients on oral bisphosphonate therapy appears to be very low; however, though the risk is small, currently millions of patients take these drugs. Therefore, recommendations for dental management of patients on oral bisphosphonate therapy were developed by an expert panel assembled by the ADA’s Council on Scientific Affairs. These panel recommendations focus on conservative surgical procedures, proper sterile technique, appropriate use of oral disinfectants and the principals of effective antibiotic therapy. There is currently no data from clinical trials evaluating dental management of patients on oral bisphosphonate therapy, and therefore, these recommendations are based on expert opinion only. A comprehensive oral evaluation is recommended for all patients about to begin therapy with oral bisphosphonates (or as soon as possible after beginning therapy).These recommendations do not address treatment of patients on i.v. bisphosphonate therapy or patients with BON. Refer to the information below regarding their treatment.
For patients on i.v. bisphosphonate therapy
It is important for dentists to be aware that while on treatment, invasive dental procedures should be avoided in patients receiving i.v. bisphosphonates, if possible. Dentists need to exercise their professional judgment, perhaps after consultation with the patient’s physician, in deciding whether invasive treatment is needed under particular clinical situations.
The prescribing information for these drugs recommends that cancer patients:
Receive a dental examination prior to initiating therapy with intravenous bisphosphonates (Aredia and Zometa), and avoid invasive dental procedures while receiving bisphosphonate treatment. For patients who develop osteonecrosis of the jaw while on bisphosphonate therapy, dental surgery may exacerbate the condition. Clinical judgment by the treating physician should guide the management plan of each patient based on an individual benefit/risk assessment.
Among tools useful to the dentist is a patient’s medical history, including medications. Dentists should be aware that patients may not relay information about receiving i.v. bisphosphonates, because these drugs are administered in oncology wards. Therefore, patients with a history of multiple myeloma, metastatic cancer, Paget’s disease, and osteoporosis may need to be questioned about receiving i.v. bisphosphonates. In addition, it may be important to know of any history of i.v. bisphosphonate administration, because these drugs have a long half-life (years).
An expert panel convened by Novartis Pharmaceuticals Corporation (the manufacturer of Zometa and Aredia) in 2004, made the following recommendations for prevention, diagnosis, and treatment of osteonecrosis of the jaw in patients on i.v. bisphosphonate therapy:
Patients should be educated on maintaining excellent oral hygiene to reduce the risk of infection.
Dentists should check and adjust removable dentures to avoid soft-tissue injury.
Routine dental cleanings should be performed with care not to inflict any soft-tissue injury.
Dental infections should be managed aggressively and nonsurgically (when possible).
Endodontic therapy is preferable to extractions; and, when necessary, coronal amputation with root canal therapy on retained roots to avoid the need for extraction.
For patients with BON
Recommendations for the treatment of patients with BON have been published and are posted on the Web site for the Journal of Oncology Practice.




